Check out our latest #whitepaper containing clinical trial data, real-world evidence, and example algorithms that detail how T2Candida can be incorporated into practice and improve care for invasive candidiasis in your own practice.
Home » Products & Technology » T2Dx Instrument
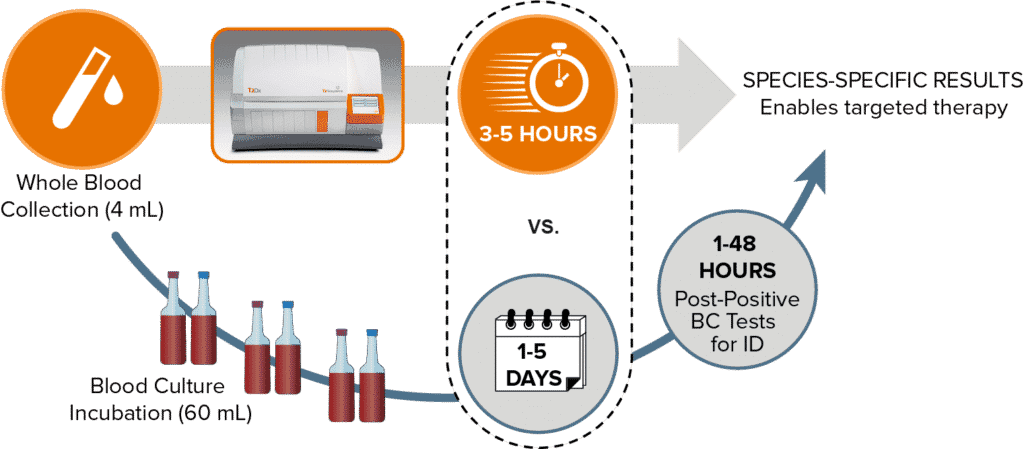
The FDA-cleared T2Dx® Instrument is a fully automated, walk away, clinical multiplex benchtop diagnostic system capable of running tests directly from whole blood.
The T2Dx Instrument serves nearly 200 hospitals worldwide and is powered by T2 Magnetic Resonance (T2MR®) technology.
Contact us to request a demo of the T2Dx Instrument!
T2 Biosystems, an emerging leader in the field of in vitro diagnostics, is dedicated to saving lives and reducing the cost of healthcare by empowering clinicians to effectively treat patients faster than ever before. T2 Biosystems is focused on addressing critical unmet needs in healthcare starting with sepsis, one of the deadliest and most expensive conditions in hospitals today.
The T2Dx Instrument, the T2Bacteria and T2Candida Panels have received marketing authorization from the U.S. Food and Drug Administration. All other T2 Biosystems products are considered investigational and for research use only.
T2 Biosystems®, T2MR®, T2Bacteria®, T2Candida®, and T2Dx® are registered trademarks of T2 Biosystems, Inc. “T2Biosystems” and the T2 Biosystems, Inc. logo design are registered trademarks or trademarks of T2Biosystems, Inc. All software and documentation is subject to T2 Biosystems, Inc. copyrights. All rights reserved. T2Direct Diagnostics™, T2HemoStat™, T2Plex™, T2Cauris™, T2Resistance™ and T2SARS-CoV-2™ are trademarks of T2 Biosystems, Inc.
© 2022 T2 Biosystems, Inc.
Please tell us where you are visiting from: United States | Outside the United States